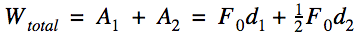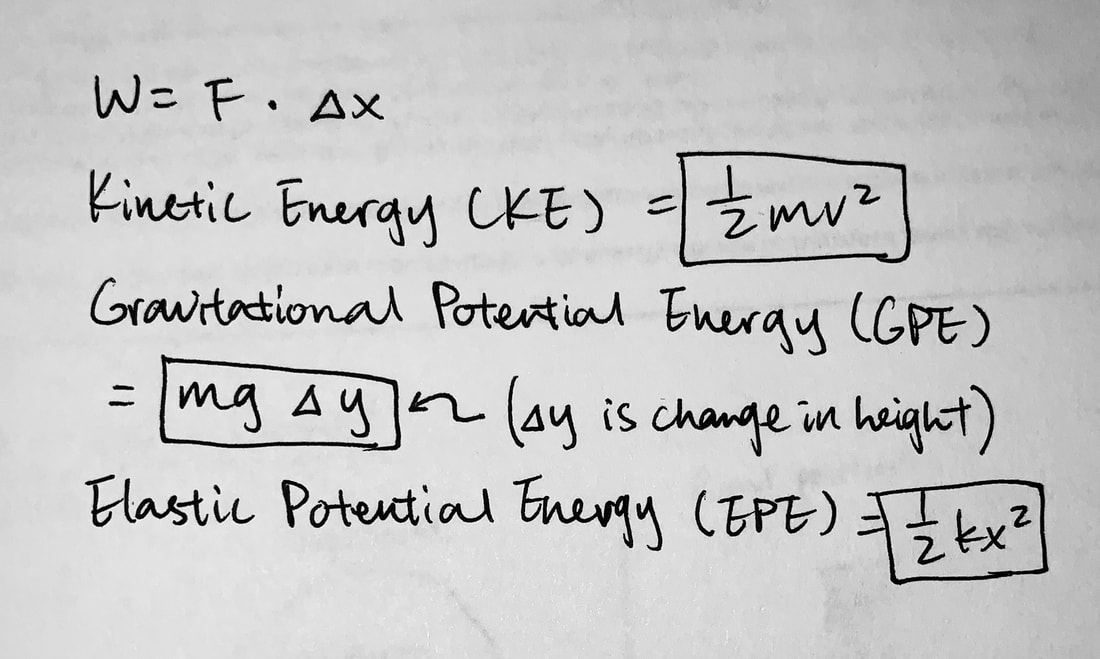KEY TERMS
Energy (E) - Measurement of the ability to do work. But really, energy is the thing that allows us to make changes. SI unit is joule (J).
Work (W) - A transfer of energy. But better, work is the change in energy by the treasfer of energy from one system to another. Scalar quantity with units of joules (J).
Joules (J) - SI unit for every. Applying a net force of 1N to an object over a displacement of 1m requires 1 J of energy. [ 1J = 1N*1m = 1 kg*m^2/s^2 ]
GENERAL EQUATIONS
Energy (E) - Measurement of the ability to do work. But really, energy is the thing that allows us to make changes. SI unit is joule (J).
Work (W) - A transfer of energy. But better, work is the change in energy by the treasfer of energy from one system to another. Scalar quantity with units of joules (J).
Joules (J) - SI unit for every. Applying a net force of 1N to an object over a displacement of 1m requires 1 J of energy. [ 1J = 1N*1m = 1 kg*m^2/s^2 ]
GENERAL EQUATIONS
|
TYPES OF ENERGY
Potential Energy - stored energy, energy caused by the object's position Kinetic Energy - the energy of motion Mechanical Energy = Potential Energy + Kinetic Energy Kinetic Energy (KE)
|
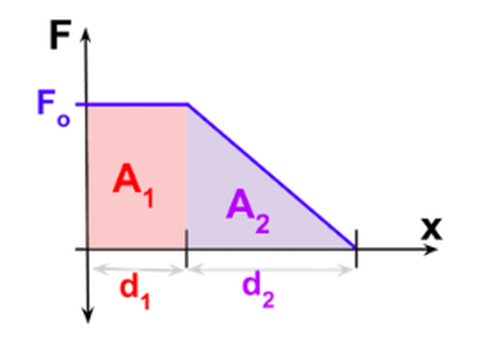
Important Equations 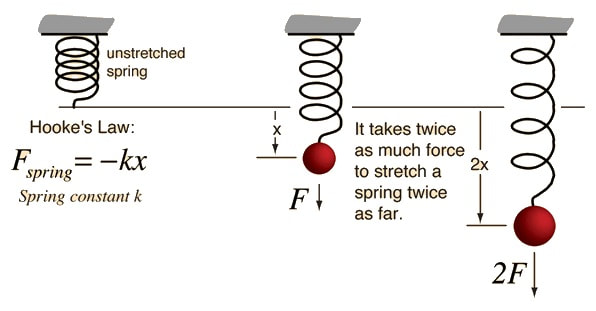
Hooke's Law
|
Law of Conservation Energy - Energy is ALWAYS Conserved
The total energy of an isolated system is constant. Energy is neither created nor destroyed, it can only be transformed from one form to another or transformed from one form to another or transferred from one system to another.
The total energy of an isolated system is constant. Energy is neither created nor destroyed, it can only be transformed from one form to another or transformed from one form to another or transferred from one system to another.
Ways of Energy Transformation - Energy can be transferred through working (W), heating (Q), or radiating (R).
Working (W)
Energy Conservation Equations
Working (W)
- Energy can be transferred by exerting an EXTERNAL push or pull on a system
- Temperature is the average amount of kinetic Energy in an object
- A temperature difference between system and surroundings causes energy to transfer from a warmer object to a cooler one
- matter loses energy by radiating light, and it gains energy when it absorbs light
Energy Conservation Equations
LOL DIAGRAMS
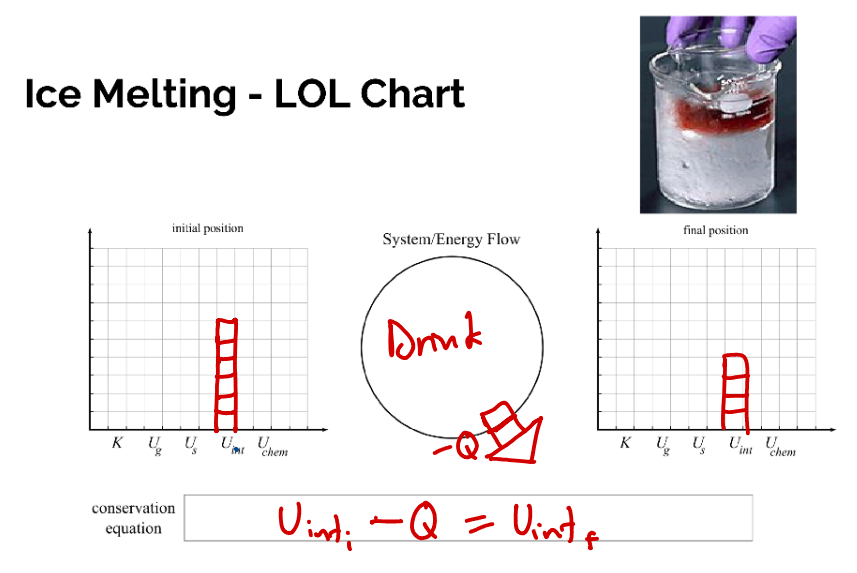 cr.Mr.Frost's class ppt
cr.Mr.Frost's class ppt
LOL DIAGRAM
example problem
 https://www.khanacademy.org/science/physics/ap-physics-1/ap-work-and-energy/power-ap/a/power-ap1
https://www.khanacademy.org/science/physics/ap-physics-1/ap-work-and-energy/power-ap/a/power-ap1
POWER
Power (P) - RATE at which work is done (or energy is transferred). SI units of Watt (W)
Watt (W) - Power equivalent to transferring 1 joule of energy per second. SI units of (kg*m^2/s^3)
Power (P) - RATE at which work is done (or energy is transferred). SI units of Watt (W)
Watt (W) - Power equivalent to transferring 1 joule of energy per second. SI units of (kg*m^2/s^3)
Power Sample Problem Solving
Widget is loading comments...